N,N-Dimethylformamide assisted hydrothermal introduction of MoS2 on ultrathin g-C3N4 layers with enhanced visible light photocatalytic hydrogen evolution activity†
Abstract
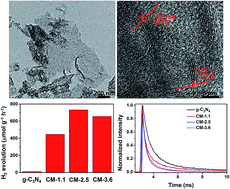
Exploring appropriate methods and agents for the fabrication of heterostructures opens up a new avenue for photocatalytic water splitting by utilizing solar power as an energy source. In this work, heterostructured photocatalysts consisting of ultrathin graphitic carbon nitride (g-C3N4) nanolayers decorated with MoS2 (g-C3N4/MoS2) were prepared through a conventional hydrothermal method with N,N-dimethylformamide (DMF) as the solvent, which prohibited the morphology damage of g-C3N4 nanosheets under high temperature and pressure. The achieved nanosized few-layer MoS2 with abundant defect sites and disordered structures was uniformly grown on the g-C3N4 nanosheets. The structures and optical properties of g-C3N4/MoS2 heterostructures were carefully characterized. By optimizing the proportion of MoS2 decoration, the g-C3N4/MoS2 heterostructure with a MoS2 content of 2.5 wt% could exhibit the highest photocatalytic H2 evolution activity under visible-light-irradiation (λ ≥ 400 nm) of 732.0 μmol g−1 h−1, which was 58.6 fold more than that of undecorated g-C3N4 (12.5 μmol g−1 h−1). Photoelectrochemical analysis and photoluminescence (PL) results demonstrated the existence of interfacial charge transfer in g-C3N4/MoS2 heterostructures, which leads to efficient separation of photogenerated electron–hole pairs. Finally, electron transfer kinetics from g-C3N4 to MoS2 were systematically investigated, and g-C3N4/MoS2-2.5 wt% (CM-2.5) presented the fastest electron injection rate (2.2 × 10−10 s−1) and highest efficiency (43.4%), which were responsible for the enhanced photocatalytic H2 evolution.
- This article is part of the themed collection: 2019 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...