Conversion of the greenhouse gas CO2 to methanol over supported intermetallic Ga–Ni catalysts at atmospheric pressure: thermodynamic modeling and experimental study†
Abstract
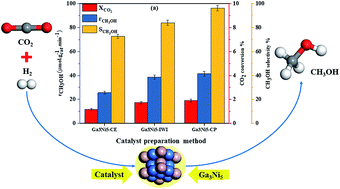
The catalytic hydrogenation of CO2 to methanol not only addresses the issue of global warming but also provides an alternative source of fuel. Herein, thermodynamic analysis of direct CO2 hydrogenation to methanol was performed by mathematical modelling over a wide range of temperatures (423 to 523 K) and CO2/H2 ratios (0.11 to 0.33). The mathematical model was derived to predict the optimized process parameters and thermodynamic limitations of this reaction at atmospheric pressure. In the experimental work, the effect of the catalyst preparation method on the structure and activity of an intermetallic Ga–Ni catalyst for CO2 hydrogenation to methanol was investigated. Three samples of the Ga–Ni alloy catalyst were prepared by condensation–evaporation, incipient wetness impregnation, and co-precipitation methods. The prepared catalysts were characterized by various surface analysis techniques, namely XRD, HRTEM, SAED, BET analysis, CO2-TPD, H2-TPR, and H2 pulse chemisorption. The catalyst prepared by the co-precipitation method (Ga3Ni5-CP) showed the maximum activity and superior physico-chemical properties compared to the catalysts prepared by other methods. Parametric study of the reaction was performed over the Ga3Ni5-CP catalyst and the reaction parameters like reactant ratio, temperature, and time on stream (stability) were optimized and found to be 0.29, 473 K, and 30 hours, respectively, at atmospheric pressure.


 Please wait while we load your content...
Please wait while we load your content...