Effective strategy for enhancing Z-scheme water splitting with the IO3−/I− redox mediator by using a visible light responsive TaON photocatalyst co-loaded with independently optimized two different cocatalysts†
Abstract
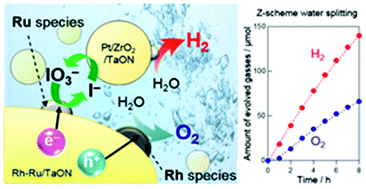
Loading an efficient cocatalyst that can selectively enhance favorable reactions is an effective way to improve the efficiency of Z-scheme water splitting with a redox mediator such as IO3−/I−. Given a recent finding on a highly active Ru(OH)xCly cocatalyst for IO3− reduction on the TaON photocatalyst, here we systematically investigate Rh, Co, and Ir-based cocatalysts for enhancing water oxidation thereby improving the total efficiency of O2 evolution on TaON with the help of the Ru(OH)xCly cocatalyst. The calcination temperature of metal cation (i.e., Rh, Co or Ir) species on TaON, which is co-loaded by Ru(OH)xCly species at a fixed temperature of 200 °C, significantly affected the rate of O2 evolution from aqueous solution containing IO3− under visible light irradiation. The rate of O2 evolution basically increased with increasing temperature at the cocatalyst loading up to 400 °C (for Ir) and 500 °C (for Rh and Co), while calcination at 600 °C drastically lowered the activity due to the detrimental effect on the TaON itself. The highest O2 evolution rate was obtained by the loading of Rh species at 500 °C, followed by co-loading of Ru(OH)xCly at 200 °C, on TaON. The results of both photocatalytic reactions and electrochemical measurements revealed that the Rh species loaded at 500 °C, which mainly consist of Rh oxides, promote water oxidation more effectively than conventional CoOx or IrOx species. The rates of H2 and O2 evolution in overall water splitting via the Z-scheme mechanism were significantly increased by the use of the TaON photocatalyst co-loaded with Rh and Ru species as the O2-evolving photocatalyst, in place of the TaON loaded with only conventional RuO2 species or previously reported TaON co-loaded with Ru(OH)xCly and Co-based species. These results indicate the effectiveness of co-loading of cocatalysts that are independently optimized for water oxidation and IO3− reduction for improving the efficiency of Z-scheme water splitting.
- This article is part of the themed collection: 3rd International Solar Fuels Conference and International Conference on Artificial Photosynthesis


 Please wait while we load your content...
Please wait while we load your content...
