Highly nanocrystalline interconnected La0.5Ca0.5CoO3−δ as an efficient bi-functional electrocatalyst for zinc–air batteries with structural and morphological evidence for ZnO mitigation†
Abstract
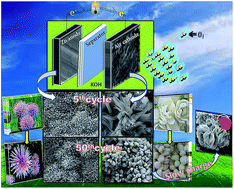
Nanocrystalline calcium-doped lanthanum cobaltate (La0.5Ca0.5CoO3−δ (nano LCCO)) synthesized by an energy efficient solution combustion method is evaluated as a cathode catalyst in zinc–air batteries. The catalyst exhibits encouraging oxygen reduction and oxygen evolution activities in an alkaline medium. Rotating disc electrode experiments show the exceptional bi-functional activity of nano LCCO (1.14 V) compared to bulk LCCO and nano LCO and is comparable to that of state of the art Pt/C and RuO2. Moreover, nano LCCO demonstrates excellent catalytic activity in zinc–air batteries at 5 mA cm−2 current density by consuming open air. A primary zinc–air battery displays a discharge capacity of 270 mA h g−1 for over 5.4 h. Rechargeable zinc–air batteries show stable electrochemical activity over 50 charge–discharge cycles and an enhanced rate performance of up to 20 mA cm−2. Post-mortem analysis of the anode and cathode catalyst by XRD and FE-SEM depicts ZnO formation and a few strategies to suppress ZnO formation have been discussed, which results in enhanced cycling stability and C-rate performance. The results presented herein make nano LCCO a highly efficient and feasible bi-functional catalyst for rechargeable zinc–air battery applications.


 Please wait while we load your content...
Please wait while we load your content...