Influence of particle size on the apparent electrocatalytic activity of LiMn2O4 for oxygen evolution†
Abstract
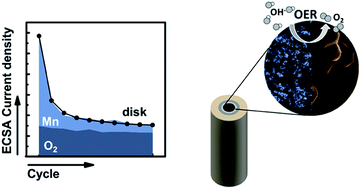
We investigate LiMn2O4 as a model catalyst for the oxygen evolution reaction (OER), because it shares the cubane structure with the active site of photosystem II. Specifically, we study the influence of different particle sizes of LiMn2O4 on the OER in a sodium hydroxide electrolyte. The product currents of manganese corrosion and oxygen evolution were obtained by rotating ring disk electrodes (RRDE). Physical characterization by various methods supports identical surface chemistry and microstructure of the pristine powders. We obtained similar oxygen current densities of 40(14) μA cmECSA−2 and 26(5) μA cmECSA−2 for micro- and nano-sized particles at 1.68 V vs. RHE. However, the total current densities differed drastically and while the micro-powder had a high disk current density of 205(2) μA cmECSA−2, its faradaic efficiency was only 25%. In contrast, the faradaic efficiency of the nanopowder was at least 75%. We hypothesize that a Mn redox process may occur in the bulk in parallel and possibly in combination with oxygen evolution on the surface based on the observed difference between the total and product current densities. Knowledge of the product currents is crucial for distinguishing the mechanisms of corrosion and catalysis and for designing better catalysts with high faradaic efficiency.


 Please wait while we load your content...
Please wait while we load your content...