Ultrasensitive recognition of AP sites in DNA at the single-cell level: one molecular rotor sequentially self-regulated to form multiple different stable conformations†
Abstract
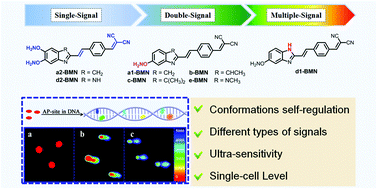
The AP site is a primary form of DNA damage. Its presence alters the genetic structure and eventually causes malignant diseases. AP sites generally present a high-speed dynamic change in the DNA sequence. Thus, precisely recognizing AP sites is difficult, especially at the single-cell level. To address this issue, we provide a broad-spectrum strategy to design a group of molecular rotors, that is, a series of nonfluorescent 2-(4-vinylbenzylidene)malononitrile derivatives (BMN-Fluors), which constantly display molecular rotation in a free state. Interestingly, after activating the relevant specific-recognition reaction (i.e., hydrolysis reaction of benzylidenemalononitrile) only in the AP-site cavity within a short time (approximately 300 s), each of these molecules can be fixed into this cavity and can sequentially self-regulate to form different stable conformations in accordance with the cavity size. The different stable conformations possess various HOMO–LUMO energy gaps in their excited state. This condition enables the AP site to emit different fluorescence signals at various wavelengths. Given the different self-regulation abilities of the conformations, the series of molecules, BMN-Fluors, can emit different types of signals, including an “OFF–ON” single-channel signal, a “ratio” double-channel signal, and even a precise multichannel signal. Among the BMN-Fluors derivatives, d1-BMN can sequentially self-regulate to form five stable conformations, thereby resulting in the emission of a five-channel signal for different AP sites in situ. Thus, d1-BMN can be used as a probe to ultrasensitively recognize the AP site with precise fluorescent signals at the single-cell level. This design strategy can be generalized to develop additional single-channel to multichannel signal probes to recognize other specific sites in DNA sequences in living organisms.


 Please wait while we load your content...
Please wait while we load your content...