Facile triflic acid-catalyzed α-1,2-cis-thio glycosylations: scope and application to the synthesis of S-linked oligosaccharides, glycolipids, sublancin glycopeptides, and TN/TF antigens†
Abstract
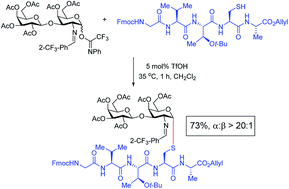
Studies of S-linked glycoconjugates have attracted growing interest because of their enhanced chemical stability and enzymatic resistance over O-glycoside counterparts. We here report a facile approach to access α-1,2-cis-S-linked glycosides using triflic acid as a catalyst to promote the glycosylation of a series of thiols with D-glucosamine, galactosamine, glucose, and galactose electrophiles. This method is broadly applicable for the stereoselective synthesis of S-linked glycopeptides, oligosaccharides and glycolipids in high yield and excellent α-selectivity. Many of the synthetic limitations associated with the preparation of these S-linked products are overcome by this catalytic method.
- This article is part of the themed collections: New reactivity in organic chemistry and 2019 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...