Enantioselective total synthesis of the unnatural enantiomer of quinine†
Abstract
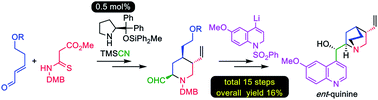
A practical enantioselective total synthesis of the unnatural (+)-quinine and (−)-9-epi-quinine enantiomers, which are important organocatalysts, is reported. The key transformation is a successive organocatalytic formal aza [3 + 3] cycloaddition/Strecker-type cyanation reaction to form an optically active tetrasubstituted piperidine derivative. This organocatalytic reaction proceeded in high yield and gave excellent enantiomeric excess with only 0.5 mol% catalyst loading. In addition, an imidate group, derived from a cyano group, was incorporated in the strategy for site-selective modification of the C4-alkyl chiral piperidine ring of quinine. Furthermore, an efficient coupling between the quinuclidine precursor and dihydroquinoline unit was achieved on a gram scale. The 15-step (LLS) synthetic protocol provided both (+)-quinine and (−)-9-epi-quinine, each with 16% overall yield.
- This article is part of the themed collections: 2021 Nobel Prize in Chemistry – Asymmetric Organocatalysis and 2019 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...