Ion-pair induced supramolecular assembly formation for selective extraction and sensing of potassium sulfate†
Abstract
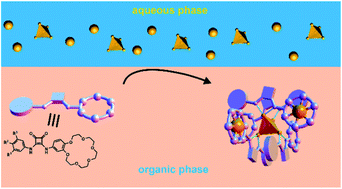
Selective extraction of sulfates in the form of alkali metal salts using charge-neutral molecular receptors is one of the holy grails of supramolecular chemistry. Herein we describe, for the first time, a squaramide-based ion pair receptor equipped with a crown ether site that is able to extract potassium sulfate from the aqueous to the organic phase (an analogous monotopic anion receptor lacking the crown ether unit lacks this ability). 1H NMR, UV-vis, DOSY-NMR, DLS, and MS experiments and the solid-state single crystal structure provided evidence of the formation of a supramolecular core–shell like assembly upon interaction of the receptor with potassium sulfate. The presence of monovalent potassium salts, in contrast, promoted the formation of simple 1 : 1 complexes. Unlike the 4 : 1 assembly, the 1 : 1 complexes are poorly soluble in organic media. This feature was utilized to overcome the Hofmeister bias and allow for selective extraction of extremely hydrophilic sulfates over lipophilic nitrate anions, which was unambiguously proved by quantitative AES and ion chromatography measurements. A simple modification of the receptor structure led to a “naked eye” optical sensor able to selectively detect sulfates under both SLE and LLE conditions.


 Please wait while we load your content...
Please wait while we load your content...