Site- and degree-specific C–H oxidation on 5-methylcytosine homologues for probing active DNA demethylation†
Abstract
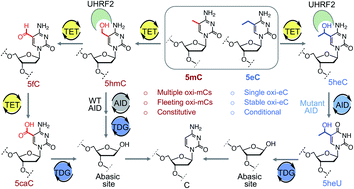
Ten-eleven translocation (TET) enzymes oxidize C–H bonds in 5-methylcytosine (5mC) to hydroxyl (5hmC), formyl (5fC) and carboxyl (5caC) intermediates en route to DNA demethylation. It has remained a challenge to study the function of a single oxidized product. We investigate whether alkyl groups other than methyl could be oxidized by TET proteins to generate a specific intermediate. We report here that TET2 oxidizes 5-ethylcytosine (5eC) only to 5-hydroxyethylcytosine (5heC). In biochemical assays, 5heC acts as a docking site for proteins implicated in transcription, imbuing this modification with potential gene regulatory activity. We observe that 5heC is resistant to downstream wild type hydrolases, but not to the engineered enzymes, thus establishing a unique tool to conditionally alter the stability of 5heC on DNA. Furthermore, we devised a chemical approach for orthogonal labeling of 5heC. Our work offers a platform for synthesis of novel 5-alkylcytosines, provides an approach to ‘tame’ TET activity, and identifies 5heC as an unnatural modification with a potential to control chromatin-dependent processes.


 Please wait while we load your content...
Please wait while we load your content...