Dye-sensitized photocathodes for oxygen reduction: efficient H2O2 production and aprotic redox reactions†
Abstract
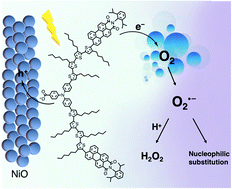
Dye-sensitized photoelectrochemical cells (DSPECs) can be used to store solar energy in the form of chemical bonds. Hydrogen peroxide (H2O2) is a versatile energy carrier and can be produced by reduction of O2 on a dye-sensitized photocathode, in which the design of dye molecules is crucial for the conversion efficiency and electrode stability. Herein, using a hydrophobic donor-double-acceptor dye (denoted as BH4) sensitized NiO photocathode, hydrogen peroxide (H2O2) can be produced efficiently by reducing O2 with current density up to 600 μA cm−2 under 1 sun conditions (Xe lamp as sunlight simulator, λ > 400 nm). The DSPECs maintain currents greater than 200 μA cm−2 at low overpotential (0.42 V vs. RHE) for 18 h with no decrease in the rate of H2O2 production in aqueous electrolyte. Moreover, the BH4 sensitized NiO photocathode was for the first time applied in an aprotic electrolyte for oxygen reduction. In the absence of a proton source, the one-electron reduction of O2 generates stable, nucleophilic superoxide radicals that can then be synthetically utilized in the attack of an available electrophile, such as benzoyl chloride. The corresponding photocurrent generated by this photoelectrosynthesis is up to 1.8 mA cm−2. Transient absorption spectroscopy also proves that there is an effective electron transfer from reduced BH4 to O2 with a rate constant of 1.8 × 106 s−1. This work exhibits superior photocurrent in both aqueous and non-aqueous systems and reveals the oxygen/superoxide redox mediator mechanism in the aprotic chemical synthesis.
- This article is part of the themed collection: Most popular 2018-2019 energy articles


 Please wait while we load your content...
Please wait while we load your content...