Zn(OTf)2-mediated annulations of N-propargylated tetrahydrocarbolines: divergent synthesis of four distinct alkaloidal scaffolds†
Abstract
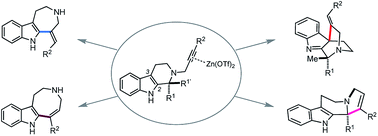
Intramolecular hydroarylations of N-propargylated tetrahydrocarbolines were efficiently mediated using a unique combination of Zn(OTf)2 with t-BuOH under neutral conditions. Use of the artificial force induced reaction method in the global reaction route mapping strategy provided insights into the Zn(OTf)2-mediated hydroarylations and the associated intriguing solvent effects of t-BuOH facilitating a protodezincation process without a Brønsted acid activator. We systematically implemented three distinct hydroarylations as well as an unanticipated α-alkenylation of a carbonyl group to obtain the four alkaloidal scaffolds 2–4, and 18. Zn(OTf)2-mediated annulation of 1c proceeded through kinetic formation of the spiroindole 3c followed by an alkenyl shift and concomitant retro-Mannich-type fragmentation to furnish azepino[4,5-b]indole 2 framework. Substituents on substrate 1 in the vicinity of the reaction sites substantially affected the mode of the divergent annulations. Judicious choices of the substituents, solvent and reaction conditions enabled programmable divergent synthesis of the four distinct skeletons.


 Please wait while we load your content...
Please wait while we load your content...