A symbiotic hetero-nanocomposite that stabilizes unprecedented CaCl2-type TiO2 for enhanced solar-driven hydrogen evolution reaction†
Abstract
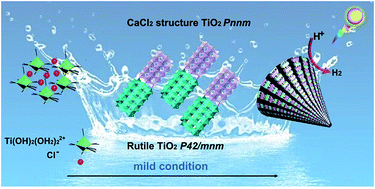
Symbiotic hetero-nanocomposites prevail in many classes of minerals, functional substances and/or devices. However, design and development of a symbiotic hetero-nanocomposite that contains unachievable phases remain a significant challenge owing to the tedious formation conditions and the need for precise control over atomic nucleation in synthetic chemistry. Herein, we report a solution chemistry approach for a symbiotic hetero-nanocomposite that contains an unprecedented CaCl2-type titania phase inter-grown with rutile TiO2. CaCl2 structured TiO2, usually occurring when bulk rutile-TiO2 is compressed at an extreme pressure of several GPa, is identified to be a distorted structure with a tilt of adjacent ribbons of the c-axis of rutile. The structural specificity of the symbiotic CaCl2/rutile TiO2 hetero-nanocomposite was confirmed by Rietveld refinement, HRTEM, EXAFS, and Raman spectra, and the formation region (TiCl4 concentration vs. reaction temperature) was obtained by mapping the phase diagram. Due to the symbiotic relationship, this CaCl2-type TiO2 maintained a high stability via tight connection by edge dislocations with rutile TiO2, thus forming a CaCl2/rutile TiO2 heterojunction with a higher reduction capacity and enhanced charge separation efficiency. These merits endow symbiotic CaCl2/rutile TiO2 with a water splitting activity far superior to that of the commercial benchmark photocatalyst, P25 under simulated sunlight without the assistance of a cocatalyst. Our findings reported here may offer several useful understandings of the mechanical intergrowth process in functional symbiotic hetero-nanocomposites for super interfacial charge separation, where interfacial dislocation appears to be a universal cause.
- This article is part of the themed collection: 2019 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...