Very short highly enantioselective Grignard synthesis of 2,2-disubstituted tetrahydrofurans and tetrahydropyrans†
Abstract
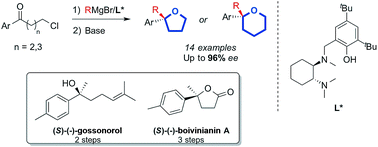
Phenones with elongated chains are shown to be excellent substrates for ligand-promoted asymmetric Grignard synthesis of tertiary alcohols. In turn this enables the simple, short and highly enantioselective (up to 96% ee) preparation of chiral 2,2-disubstituted THFs and THPs. Thus, asymmetric addition of Grignard reagents to γ-chlorobutyrophenones and δ-chlorovalerophenones takes place in the presence of a chiral diaminocyclohexyl-derived tridentate ligand and subsequent base-promoted intramolecular cyclisation occurs with complete retention of asymmetry. As examples of the methodology, we report the shortest syntheses of gossonorol, γ-ethyl-γ-phenylbutyrolactone and δ-methyl-δ-tolylvalerolactone, the joint-shortest and flexible synthesis of boivinianin A and the shortest formal syntheses of boivinianin B and yingzhaosu C.


 Please wait while we load your content...
Please wait while we load your content...