Symmetry breaking and the turn-on fluorescence of small, highly strained carbon nanohoops†
Abstract
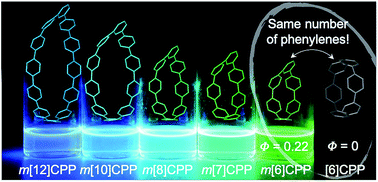
[n]Cycloparaphenylenes, or “carbon nanohoops,” are unique conjugated macrocycles with radially oriented π-systems similar to those in carbon nanotubes. The centrosymmetric nature and conformational rigidity of these molecules lead to unusual size-dependent photophysical characteristics. To investigate these effects further and expand the family of possible structures, a new class of related carbon nanohoops with broken symmetry is disclosed. In these structures, referred to as meta[n]cycloparaphenylenes, a single carbon–carbon bond is shifted by one position in order to break the centrosymmetric nature of the parent [n]cycloparaphenylenes. Advantageously, the symmetry breaking leads to bright emission in the smaller nanohoops, which are typically non-fluorescent due to optical selection rules. Moreover, this simple structural manipulation retains one of the most unique features of the nanohoop structures—size dependent emissive properties with relatively large extinction coefficients and quantum yields. Inspired by earlier theoretical work by Tretiak and co-workers, this joint synthetic, photophysical, and theoretical study provides further design principles to manipulate the optical properties of this growing class of molecules with radially oriented π-systems.
- This article is part of the themed collections: The ChemRxiv Collection and 2019 International Open Access Week Collection


 Please wait while we load your content...
Please wait while we load your content...