Dinuclear HgII tetracarbene complex-triggered aggregation-induced emission for rapid and selective sensing of Hg2+ and organomercury species†‡
Abstract
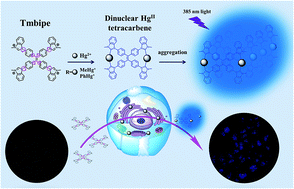
Rapid, reliable and highly selective detection of mercury species, including Hg2+ ions and organomercury, is of significant importance for environmental protection and human health. Herein, a new fluorescent dye 1,1,2,2-tetrakis[4-(3-methyl-1H-benzimidazol-1-yl)phenyl ethylene tetraiodide (Tmbipe) with aggregation-induced emission (AIE) potential was prepared and characterized. The presence of four positively charged methylated benzimidazole groups endows Tmbipe with excellent water solubility and almost undetectable background fluorescence. However, it can coordinate with two Hg2+ ions or two organomercury molecules (e.g. methylmercury and phenylmercury) to form a planar dinuclear HgII tetracarbene complex, which can then self-aggregate to turn on AIE fluorescence. Such a fluorescence turn-on process can be completed in 3 min. In addition, synergic rigidification of the tetraphenylethylene-bridged Tmbipe molecule by mercury-mediated chelate ring formation and subsequent aggregation results in obviously higher fluorescence enhancement than that given by the single aggregation-induced one. Low background, high fluorescence enhancement and rapid response time make Tmbipe a good fluorescent probe for reliable, sensitive and highly selective quantitation of both inorganic and organic mercury species. This probe was also demonstrated to work well for identification of mercury species accumulation in living cells.
- This article is part of the themed collection: Celebrating 100 Years of Chemistry at Nankai University


 Please wait while we load your content...
Please wait while we load your content...