Facile access to functionalized chiral secondary benzylic boronic esters via catalytic asymmetric hydroboration†
Abstract
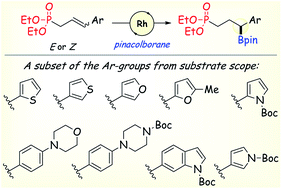
Allylic and homoallylic phosphonates bearing an aryl or heteroaryl substituent at the γ- or δ-position undergo rhodium-catalyzed asymmetric hydroboration by pinacolborane to give functionalized chiral secondary benzylic boronic esters in yields up to 86% and enantiomer ratios up to 99 : 1. Compared to minimally-functionalized terminal and 1,1-disubstituted vinyl arenes, there are relatively few reports of efficient catalytic asymmetric hydroboration (CAHB) of more highly functionalized internal alkenes. Phosphonate substrates bearing a variety of common heterocyclic ring systems, including furan, indole, pyrrole and thiophene derivatives, as well as those bearing basic nitrogen substituents (e.g., morpholine and pyrazine) are tolerated, although donor substituents positioned in close proximity of the alkene can influence the course of the reaction. Stereoisomeric (E)- and (Z)-substrates afford the same major enantiomer of the borated product. Deuterium-labelling studies reveal that rapid (Z)- to (E)-alkene isomerization accounts for the observed (E/Z)-stereoconvergence during CAHB. The synthetic utility of the chiral boronic ester products is illustrated by stereospecific C–B bond transformations including stereoretentive electrophile promoted 1,2-B-to-C migrations, stereoinvertive SE2 reactions of boron-ate complexes with electrophiles, and stereoretentive palladium- and rhodium-catalyzed cross-coupling protocols.


 Please wait while we load your content...
Please wait while we load your content...