Toward ideal carbon dioxide functionalization
Abstract
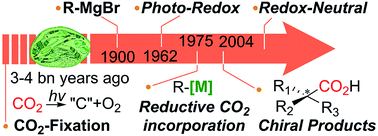
This Perspective recapitulates recent developments of carbon dioxide utilization in carbon–carbon bond formation reactions, with an intention of paving a way toward sustainable CO2-functionalization and its tangible applications in synthetic chemistry. CO2 functionalization reactions possess intrinsic drawbacks: the high kinetic inertness and thermodynamic stability of CO2. Numerous procedures for CO2 utilization depend on energy-intensive processes (i.e. high pressure and/or temperature), often solely relying on reactive substrates, hampering its general applications. Recent efforts thus have been dedicated to catalytic CO2-utilization under ambient reaction conditions, however, it is still limited to a few activation modes and the use of reactive substrates. Herein, ideal CO2-functionalization with particular emphasis on sustainability will be discussed based on the following sub-categories; (1) metal-catalyzed ‘reductive’ carboxylation reaction of halides, olefins and allyl alcohols, (2) photochemical CO2-utilization, (3) redox-neutral CO2-functionalization, and (4) enantioselective catalysis incorporating CO2 to form C–CO2 bonds (excluding strain mediated reactions with epoxide- and aziridine-based substrates). Recent progress in these fields will be discussed with the proposed reaction mechanisms and selected examples, highlighting redox-neutral, umpolung, and asymmetric carboxylation to postulate ideal CO2 functionalization reactions to be developed in the near future.
- This article is part of the themed collections: Most popular 2019-2020 review articles, Most popular 2019-2020 organic chemistry articles and How can chemistry adapt to a low carbon future


 Please wait while we load your content...
Please wait while we load your content...