Light emission enhancement by supramolecular complexation of chemiluminescence probes designed for bioimaging†
Abstract
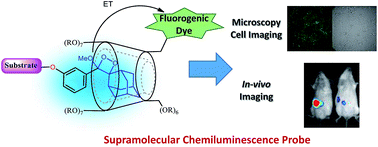
Chemiluminescence offers advantages over fluorescence for bioimaging, since an external light source is unnecessary with chemiluminescent agents. This report demonstrates the first encapsulation of chemiluminescence phenoxy-adamantyl-1,2-dioxetane probes with trimethyl β-cyclodextrin. Clear proof for the formation of a 1 : 1 host–guest complex between the adamantyl-1,2-dioxetane probe and trimethyl β-cyclodextrin was provided by mass spectroscopy and NMR experiments. The calculated association constant of this host–guest system, 253 M−1, indicates the formation of a stable inclusion complex. The inclusion complex significantly amplified the light emission intensity relative to the noncomplexed probe under physiological conditions. Complexation of adamantyl-dioxetane with fluorogenic dye-tethered cyclodextrin resulted in light emission through energy transfer to a wavelength that corresponds to the fluorescent emission of the conjugated dye. Remarkably, the light emission intensity of this inclusion complex was approximately 1500-fold higher than that of the non-complexed adamantyl-dioxetane guest. We present the first demonstration of microscopic cell images obtained using a chemiluminescence supramolecular dioxetane probe and demonstrate the utility of these supramolecular complexes by imaging of enzymatic activity and bio-analytes in vitro and in vivo. We anticipate that the described chemiluminescence supramolecular dioxetane probes will find use in various biological applications.
- This article is part of the themed collections: Near-infrared (NIR) luminescent probes for bioimaging and biosensing and Most popular 2019-2020 supramolecular chemistry articles


 Please wait while we load your content...
Please wait while we load your content...