Enantioselective and site-specific copper-catalyzed reductive allyl–allyl cross-coupling of allenes†
Abstract
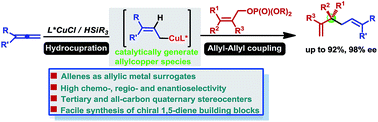
A copper-catalyzed asymmetric reductive allyl–allyl cross-coupling reaction of allenes with allylic phosphates wherein allenes were used as allylmetal surrogates has been achieved for the first time. This protocol provides an efficient and straightforward route to optically active 1,5-dienes in a highly enantioselective and site-specific fashion. Furthermore, all-carbon quaternary stereogenic centers could also be constructed with this protocol. The versatility of the products is demonstrated through a diverse array of further transformations of the enantioenriched 1,5-dienes.


 Please wait while we load your content...
Please wait while we load your content...