Electrocatalytic reduction of low concentration CO2†
Abstract
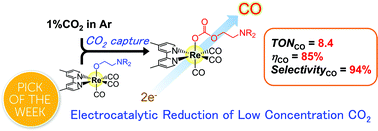
Utilization of low concentration CO2 contained in the exhaust gases from various industries and thermal power stations without the need for energy-consuming concentration processes should be an important technology for solving global warming and the shortage of fossil resources. Here we report the direct electrocatalytic reduction of low concentration CO2 by a Re(I)-complex catalyst that possesses CO2-capturing ability in the presence of triethanolamine. The reaction rate and faradaic efficiency of CO2 reduction were almost the same when using Ar gas containing 10% CO2 or when using pure CO2 gas, and the selectivity of CO formation was very high (98% at 10% CO2). At a concentration of 1% CO2, the Re(I) complex still behaved as a good electrocatalyst; 94% selectivity of CO formation and 85% faradaic efficiency were achieved, and the rate of CO formation was 67% compared to that when using pure CO2 gas. The electrocatalysis was due to the efficient insertion of CO2 into the Re(I)–O bond in fac-[Re(dmb)(CO)3{OC2H4N(C2H4OH)2}] (dmb = 4,4′-dimethyl-2,2′-bipyridine).
- This article is part of the themed collection: 2018 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...