Electrocatalytic water oxidation over AlFe2B2†
Abstract
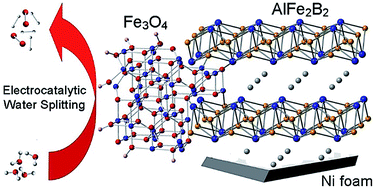
We report excellent electrocatalytic performance by AlFe2B2 in the oxygen-evolution reaction (OER). The inexpensive catalytic material, prepared simply by arc-melting followed by ball-milling, exhibits high stability and sustained catalytic performance under alkaline conditions. The overpotential value of 0.24 V observed at the current density of 10 mA cm−2 remained constant for at least 10 days. Electron microscopy and electron energy loss spectroscopy performed on the initial ball-milled material and on the material activated under electrocatalytic conditions suggest that the catalytic mechanism involves partial leaching of Al from the layered structure of AlFe2B2 and the formation of Fe3O4 nanoclusters on the exposed [Fe2B2] layers. Thus, the AlFe2B2 structure serves as a robust supporting material and, more importantly, as a pre-catalyst to the in situ formed active electrocatalytic sites. Comparative electrochemical measurements demonstrate that the electrocatalytic performance of the AlFe2B2-supported Fe3O4 nanoclusters substantially exceeds the results obtained with unsupported nanoparticles of Fe3O4, FeB, or such benchmark OER catalysts as IrO2 or RuO2. The excellent catalytic performance and long-term stability of this system suggests that AlFe2B2 can serve as a promising and inexpensive OER electrocatalyst.


 Please wait while we load your content...
Please wait while we load your content...