Photolysis of cell-permeant caged inositol pyrophosphates controls oscillations of cytosolic calcium in a β-cell line†
Abstract
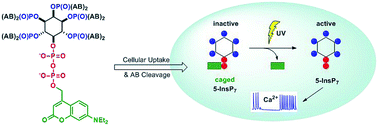
Among many cellular functions, inositol pyrophosphates (PP-InsPs) are metabolic messengers involved in the regulation of glucose uptake, insulin sensitivity, and weight gain. However, their mechanisms of action are still poorly understood. So far, the influence of PP-InsPs on cellular metabolism has been studied by overexpression or knockout/inhibition of relevant metabolizing kinases (IP6Ks, PPIP5Ks). These approaches are, inter alia, limited by time-resolution and potential compensation mechanisms. Here, we describe the synthesis of cell-permeant caged PP-InsPs as tools to rapidly modulate intracellular levels of defined isomers of PP-InsPs in a genetically non-perturbed cellular environment. We show that caged prometabolites readily enter live cells where they are enzymatically converted into still inactive, metabolically stable, photocaged PP-InsPs. Upon light-triggered release of 5-PP-InsP5, the major cellular inositol pyrophosphate, oscillations of intracellular Ca2+ levels in MIN6 cells were transiently reduced to spontaneously recover again. In contrast, uncaging of 1-PP-InsP5, a minor cellular isomer, was without effect. These results provide evidence that PP-InsPs play an active role in regulating [Ca2+]i oscillations, a key element in triggering exocytosis and secretion in β-cells.


 Please wait while we load your content...
Please wait while we load your content...