Stereoselective organocatalyzed glycosylations – thiouracil, thioureas and monothiophthalimide act as Brønsted acid catalysts at low loadings†
Abstract
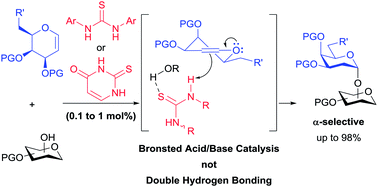
Thiouracil catalyzes stereoselective glycosylations with galactals in loadings as low as 0.1 mol%. It is proposed that in these glycosylations thiouracil, monothiophthalimide, and the previously reported catalyst, Schreiner's thiourea, do not operate via a double H-bonding mechanism but rather by Brønsted acid/base catalysis. In addition to the synthesis of 2-deoxyglycosides and glycoconjugates, we report the first organocatalytic synthesis of 1,1′-linked trehalose-type sugars.


 Please wait while we load your content...
Please wait while we load your content...