Kinetic study of TBD catalyzed δ-valerolactone polymerization using a gas-driven droplet flow reactor†
Abstract
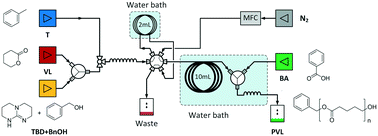
The kinetics of aliphatic polyester synthesis is critical to understanding the mechanism of ring-opening polymerization and making the reaction more controllable. In this paper, we introduce a gas-driven droplet flow reactor to determine the reaction kinetics of 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) catalyzed δ-valerolactone polymerization. Only 2 mL reaction solution was used for each test, which significantly reduced the work in preparing the anhydrous reactants. The experimental results indicated that the orders of the catalyst and the alcohol in the kinetic model are close to 1 and 0, respectively, and therefore the rate-determining step was the activation reaction between the monomer and catalyst. Then, the activated monomer quickly reacts with the end hydroxyl in the polymer chain to finish the reaction. In the end, we calculated the activation energy of the rate-determining reaction, which is only 7.302 ± 0.047 kJ mol−1.
- This article is part of the themed collection: 2019 Reaction Engineering in China


 Please wait while we load your content...
Please wait while we load your content...