Metal-free catalytic oxidation of benzylic alcohols for benzaldehyde†
Abstract
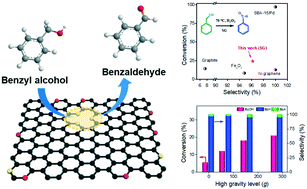
Sulfurized graphene (SG) was employed as a metal-free catalyst for the liquid-phase catalytic oxidation of benzyl alcohol (BzOH) in the existence of hydrogen peroxide for the first time. The conversion efficiencies of BzOH at different reaction conditions were studied. SG was demonstrated to be among the best metal-free catalysts for this reaction in conversional stirred tank reactors with BzOH conversion of 18.2% (2 mg catalyst, 3 h, T = 80 °C). A high-gravity rotating packed bed reactor was used for process intensification during the metal-free catalytic oxidation, and BzOH conversion increased from 5.56% to 20.86%. Both experimental and computational fluid dynamics simulations indicate that a high-gravity level can provide more turbulent kinetic energy and faster surface renewal rate to improve the catalytic efficiency. This study opened up the possibility of combining metal-free catalysts and high-gravity technology for the green synthesis of chemicals.
- This article is part of the themed collection: 2019 Reaction Engineering in China


 Please wait while we load your content...
Please wait while we load your content...