Origins of complex solvent effects on chemical reactivity and computational tools to investigate them: a review
Abstract
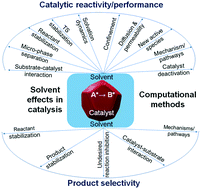
Solvents are crucial components in specialty chemical and pharmaceutical industries and in electrochemical and photoelectrochemical processes, and are increasingly being used in catalytic reactions. Solvents significantly influence the kinetics and thermodynamics of reactions and can alter product selectivity markedly. While such solvent effects are observed routinely, identification of the root causes of such effects is less frequent. Solvents can influence reaction rates, conversion and product selectivity by 1) directly participating in the reaction steps and opening alternate reaction pathways, 2) competing with the reactant for interaction with the catalysts, 3) changing the relative stabilization of the reactant, the transition state (TS) and/or the product, 4) altering intra-pore diffusion characteristics in porous catalysts, 5) exhibiting entropic confinement effects altering free energy barriers of reactions, 6) changing the solubility of different components in the reaction mixture, and 7) inhibiting undesired reactions. Their indirect influences may be due to 1) changes brought on to active sites on catalysts and 2) altered structure/stability of catalysts. This article discusses these fundamental reasons behind observed solvent effects with suitable examples. Advances in computational chemistry have led to the development of multiple tools and techniques, considering solvents either as implicit or as explicit molecules, providing molecular insights into complex solvent effects in catalysis. This article provides an overview of some of these methods with suitable examples to demonstrate their application and potential. This mapping of the solvent effects and their origins is believed to aid in the rational selection of solvents for catalytic reactions. The description of the computational tools, their application and their potential is likely to encourage widespread use of these techniques to investigate solvent effects.
- This article is part of the themed collection: Reaction Chemistry & Engineering Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...