Kinetic modelling for the hydrogenolysis of bio-glycerol in the presence of a highly selective Cu–Ni–Al2O3 catalyst in a slurry reactor
Abstract
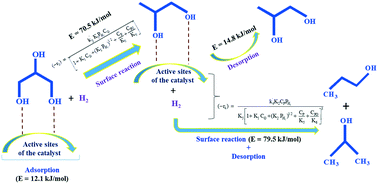
A kinetic study on the liquid phase hydrogenolysis of glycerol was carried out in a slurry reactor in the presence of a highly selective Cu–Ni–Al2O3 catalyst at different reaction temperatures (180–220 °C) and pressures (3–6 MPa), and very high selectivity was achieved (∼95%) towards 1,2-propanediol (1,2-PDO). A series of reaction schemes for the conversion of glycerol to 1,2-PDO followed by the hydrogenolysis of 1,2-PDO to propanol was considered to develop the kinetic model. Initially, the power-law model was fitted followed by a modified power-law model considering 1,2-PDO and propanol (1-PO + 2-PO) as the main reaction products. To understand the intrinsic kinetic behaviour, a more realistic heterogeneous kinetic model based on a combined Langmuir–Hinshelwood–Hougen–Watson (LHHW) and Eley–Rideal (ER) approach was developed. The numerical solution for the model equation was computed using ode23 solver in MATLAB combined with the genetic algorithm. The results demonstrated that the experimental concentrations were well-fitted with the model-predicted concentration. The calculated activation energy was 70.5 kJ mol−1 for the conversion of glycerol to 1,2-PDO and 79.5 kJ mol−1 for the production of PO from 1,2-PDO. The parity plots of the experimental and model simulated concentration of the reactant and products were fitted very well.


 Please wait while we load your content...
Please wait while we load your content...