Linker length in fluorophore–cholesterol conjugates directs phase selectivity and cellular localisation in GUVs and live cells†
Abstract
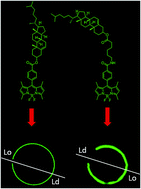
Lipid membrane fluorescent probes that are both domain-selective and compatible with demanding microscopy methods are crucial to elucidate the presence and function of rafts and domains in cells and biophysical models. Whereas targeting fluorescent probes to liquid-disordered (Ld) domains is relatively facile, it is far more difficult to direct probes with high selectivity to liquid-ordered (Lo) domains. Here, a simple, one-pot approach to probe–cholesterol conjugation is described using Steglich esterification to synthesise two identical BODIPY derivatives that differ only in the length of the aliphatic chain between the dye and cholesterol. In the first, BODIPY-Ar-Chol, the probe and cholesterol were directly ester linked and in the second BODIPY-Ahx-Chol, a hexyl linker separated probe from cholesterol. Uptake and distribution of each probe was compared in ternary, phase separated giant unilamellar vesicles (GUVs) using a commercial Ld marker as a reference. BODIPY-Ar-Chol targets almost exclusively the Ld domains with selectivity of >90% whereas by contrast introducing the C6 linker between the probe and cholesterol drove the probe to Lo with excellent selectivity (>80%). The profound impact of the linker length extended also to uptake and distribution in live mammalian cells. BODIPY-Ahx-Chol associates strongly with the plasma membrane where it partitioned preferably into opposing micron dimensioned do-mains to a commercial Ld marker and its concentration at the membrane was reduced by cyclodextrin treatment of the cells. By contrast the BODIPY-Ahx-Chol permeated the membrane and localised strongly to lipid droplets within the cell. The data demonstrates the profound influence of linker length in cholesterol bioconjugates in directing the probe.


 Please wait while we load your content...
Please wait while we load your content...