Visible-light-mediated difluoromethylphosphonation of alkenes for the synthesis of CF2P-containing heterocycles†
Abstract
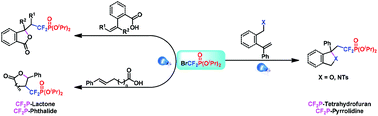
Difluoromethylphosphonate, as an in vivo stable phosphate bio-isostere, is an important organofluorophosphine block. In this paper, an efficient and general protocol for the construction of CF2P-containing heterocycles via visible-light-mediated nucleophilic cyclization of unsaturated carboxylic acids, alcohol and sulfonamides has been developed. This reaction can be carried out smoothly under mild conditions with less catalyst loading, producing several types of CF2P-containing heterocycles, including CF2P-lactone, CF2P-phthalide, CF2P-tetrahydrofuran and CF2P-pyrrolidine.


 Please wait while we load your content...
Please wait while we load your content...