Four intricately caged polycyclic polyprenylated acylphloroglucinols from Garcinia multiflora fruits†
Abstract
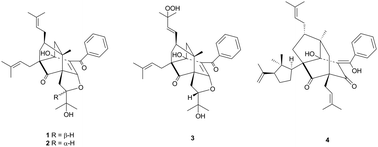
Garmultinones A–C (1–3), three polycyclic polyprenylated acylphloroglucinols (PPAPs) with a unique caged tricyclo[3.3.1.1.4,8]decane skeleton, and one PPAP garmultinone D (4) with the tricyclo[4.3.1.03,7]decane skeleton bearing the 2,2-dimethyl-3-isopropenylcyclopentyl ring were isolated from the fruits of Garcinia multiflora. Their structures and absolute configurations were determined by extensive NMR analysis and computational calculation methods. The anti-inflammatory effects of these isolates were evaluated by a lipopolysaccharide (LPS)-stimulated nitric oxide (NO) production model in RAW264.7 macrophage cells. The possible biogenetic pathway of compounds 1–4 through the intramolecular [4 + 2] cycloadditions of monocyclic polyprenylated acylphloroglucinols (MPAPs) was also proposed.


 Please wait while we load your content...
Please wait while we load your content...