Unusual structural changes as a result of weathering benzofuran-based diarylethenes in simulated sunlight†
Abstract
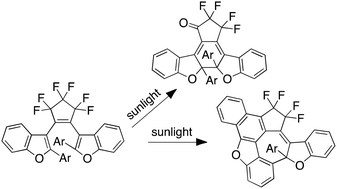
While the products that result from photochemical degradation of thiophene-based diarylethenes have been documented, those from the benzofuran counterparts have been less extensively studied. In this article, we present two unusual and yet to be reported degradation products of a dialkoxybenzofuran derivative, characterized by a combination of spectroscopy techniques and X-ray crystallography. We show that these products are generated by due to the specific substitution position of the alkoxy groups on the benzofuran heterocycle. We also propose reasonable mechanisms for the degradation pathways and products formation, which are supported by equivalent studies using isomeric monoalkoxy versions.


 Please wait while we load your content...
Please wait while we load your content...