An empirical study of amide–heteroarene π-stacking interactions using reversible inhibitors of a bacterial serine hydrolase†
Abstract
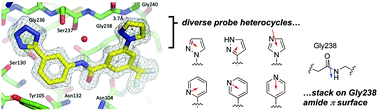
Compared to aryl–aryl π-stacking interactions, the analogous stacking of heteroarenes on amide π systems is less well understood and vastly underutilized in structure-based drug design. Recent theoretical studies have delineated the important geometric coordinates of the interaction, some of which have been confirmed with synthetic model systems based on Rebek imides. Unfortunately, a broadly useful and tractable protein–ligand model system of this interaction has remained elusive. Here we employed a known inhibitor scaffold to study π-stacking of diverse heteroarene substituents on the amide face of Gly238 in the cephalosporinases CTX-M-14 and CTX-M-27. Biochemical inhibition constants (Ki) and biophysical binding constants (Kd) were determined for nineteen new analogues against both enzymes, while multiple high-resolution co-crystal structures revealed remarkably consistent placement of the probe heteroarene on Gly238. The data presented support the predicted importance of opposing dipoles in amide–heteroarene interactions and should be useful for evaluating other theoretical predictions concerning these interactions.
- This article is part of the themed collection: In celebration of Julius Rebek’s 75th Birthday


 Please wait while we load your content...
Please wait while we load your content...