Role of double interfaces in inspiring energy storage devices in CC@Ni(OH)Cl@NiO flexible electrodes†
Abstract
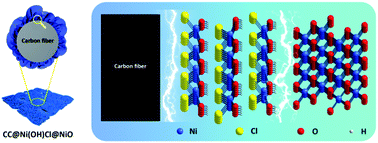
Interfacial engineering is highly promising in the research field of flexible energy storage devices. Massive efforts have confirmed the beneficial effects of controlled interfaces in enhancing energy storage. Despite the extensive studies on constructing multilevel interfaces in the fabrication of flexible electrodes, a deep understanding of the role of interfaces from the electrochemical perspective is lacking, particularly the operational feasibility in extreme conditions. Herein, we designed a carbon cloth (CC) based double-interface CC@Ni(OH)Cl@NiO by a solvothermal, chemical bath, and subsequent annealing method. We obtained both remarkable areal capacitance (8290 mF cm−2 at 30 mA cm−2) and excellent cycling stability (73.9% capacitance retention after 1000 cycles), approximately double than those for the single-interface CC@Ni(OH)Cl. Correspondingly, ultrahigh current tolerance was endowed with a stable charge–discharge capacitance of 3580 mF cm−2 at 120 mA cm−2. A flexible quasi-solid-state asymmetric supercapacitor device, CC@Ni(OH)Cl@NiO//graphene, was assembled, which achieved favorable capacitive ability and splendid flexibility as well as mechanical stability. Through the synchrotron radiation technique, we demonstrated that synergistic double interfaces afford the benefits of stable Ni–O covalency during the electrochemical cycles, and meanwhile bring about more structural distortion and active electronic behavior on the surface state, which simultaneously stimulated capacitive ability and stability. Importantly, our study reveals a positive role of double interfaces from the electrochemical perspective of flexible electrodes for widespread energy storage in the future.


 Please wait while we load your content...
Please wait while we load your content...