A series of red iridium(iii) complexes using flexible dithiocarbamate derivatives as ancillary ligands for highly efficient phosphorescent OLEDs†
Abstract
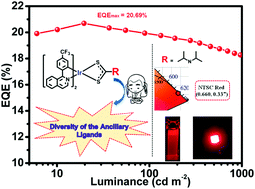
Five red cyclometalated iridium(III) complexes containing the unique four-membered ring Ir–S–C–S backbone with different dithiocarbamate derivatives as ancillary ligands and 1-(4-(trifluoromethyl)phenyl)isoquinoline (4tfmpiq) as the main ligand were synthesized at room temperature within ten minutes. The emission color (λpeak = 607–619 nm) and photoluminescence quantum efficiency (PLQY) (ΦP = 36.6–63.0%) can be effectively tuned by introducing different electron – donating substituents such as diisopropylamine (dipdtc), diphenylamine (dpdtc), bis(4-(tert-butyl)phenyl)amine (tBudpdtc), carbazole (Czdtc) and 3,6-di-tert-butyl-9H-carbazole (tBuCzdtc) into the dithiocarbamate ancillary ligands. Employing (4tfmpiq)2Ir(dipdtc) as the emitter, the organic light emitting diodes (OLEDs) with double emitting layers exhibit good performances with a maximum current efficiency (ηc,max) of 18.67 cd A−1, an EQEmax of 20.69% and low efficiency roll-off with an EQE of 18.11% at the practical luminance of 1000 cd m−2. The Commision Internationale de L’Eclairage coordinates of (0.660, 0.337) are quite close to the National Television System Comittee (NTSC) standard red emission. These results demonstrate that by introducing different electron – donating substituents into the dithiocarbamate ligands, the diversity of the S–C–S ligands is greatly enriched for red Ir(III) complexes and the corresponding OLEDs.
- This article is part of the themed collection: 2019 Materials Chemistry Frontiers HOT articles


 Please wait while we load your content...
Please wait while we load your content...