Superalkali ligands as a building block for aromatic trinuclear Cu(i)–NHC complexes†
Abstract
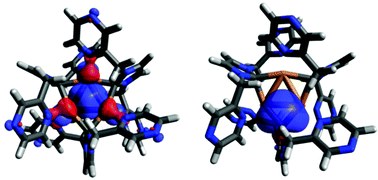
Imidazole and benz-imidazole based different NHC ligands have been designed to synthesize a Cu(I)@NHC complex. Calculated vertical electron affinity suggests that all the ligands belong to a special type of molecule, superalkali, and shows a very good non linear optical activity. All of them form Cu(I)@NHC complexes having a trinuclear Cu3 core. Canonical Molecular Orbital contribution towards nucleus-independent chemical shift values suggest that the Cu3 ring is both σ- and π-aromatic. Aromaticity and other reactivity parameters like electrophilicity and hardness change with different ligand environments. It has been found that pyrazine based NHC synthesizes a more reactive complex than pyridine. Natural localized molecular orbitals of these complexes suggest that they have sp2 hybridization like BH3.


 Please wait while we load your content...
Please wait while we load your content...
