Hollow cobalt sulfide for highly efficient uranium adsorption from aqueous solutions†
Abstract
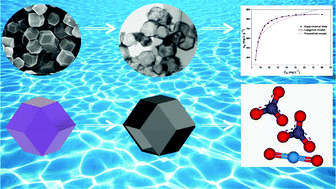
With the increasing and sustainable development of nuclear industry, it is of significant importance to develop efficient adsorbents with high adsorption capacity and selectivity for uranium. In this work, hollow Co3S4 was explored to remove radioactive uranium from aqueous solution. The equilibrium adsorption data were fitted well by the Langmuir model and the maximum adsorption capacity of 863.71 mg g−1 was obtained at pH ∼5.0. The adsorption kinetics result revealed that hollow Co3S4 could adsorb uranium very rapidly via a diffusion process. The study of its reusability by applying adsorption–desorption cycles was also conducted and showed that the hollow Co3S4 possesses good stability with a negligible capacity decrease after four cycles. In addition, the hollow adsorbent exhibited high removal efficiency in both deionized water and tap water, which indicates a promising potential for applications in the efficient separation of uranium from aqueous solutions.


 Please wait while we load your content...
Please wait while we load your content...