Copper(ii) facilitated decarboxylation for the construction of pyridyl–pyrazole skeletons†
Abstract
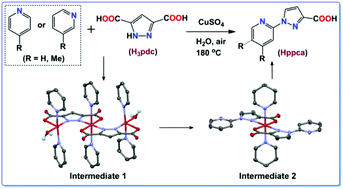
A highly simple and practical method was explored for the synthesis of pyridyl–pyrazole carboxylic compounds in one step by Cu(II) facilitated decarboxylation of 3,5-pyrazole-dicarboxylic acid (H3pdc) in pyridine–water. The activation of pyridine was induced by a hydroxyl radical, originating from the homolytic cleavage of the pz-COO− bond, and thereafter transfer of the radical to water. Mechanistic studies showed that the elongation of the CuII–Ocarboxyl bond in the side cores of [Cu3(pdc)2(py)6(H2O)2] (1) promoted the generation of reactive radicals through decarboxylation. A possible reaction mechanism was proposed, involving a radical-induced pyridine C–H bond activation and a copper-facilitated pyridyl–pyrazole C–N bond formation.


 Please wait while we load your content...
Please wait while we load your content...