Fe/N-doped carbon nanofibers with Fe3O4/Fe2C nanocrystals enchased as electrocatalysts for efficient oxygen reduction reaction†
Abstract
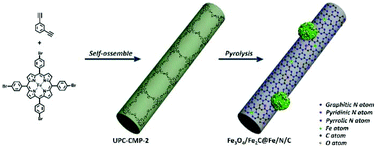
Designing efficient non-noble metal catalysts with good oxygen reduction activity is highly desirable but challenging. Herein, we demonstrate the synthesis of Fe-based carbon materials with multi active sites of Fe–Nx, Fe3O4 and Fe2C for oxygen reduction through facile pyrolysis of a Fe–porphyrin conjugated microporous polymer. The nanofiber structure of the polymer is retained with in situ Fe and N heteroatom doping and Fe3O4/Fe2C composite nanocrystals enchased in the walls of carbon fibers. Synergism between the N-doped carbon nanofibers and the in situ formed active sites of Fe–Nx, Fe2C and Fe3O4 facilitates the electrocatalytic performance of the as-synthesized catalysts. The catalyst obtained from 800 °C pyrolysis exhibits prominent oxygen reduction catalytic performance in 0.1 M KOH with an onset potential of 0.91 V, a half-wave potential of 0.81 V, and a small Tafel slope of 74 mV dec−1. Its limiting current density of −5.3 mA cm−2, excellent durability and resistance to methanol surpass the commercial 20% Pt/C under identical conditions. Thus, this work may provide a guideline for the design and synthesis of multi active nonprecious metal electrocatalysts for future fuel–cell applications.


 Please wait while we load your content...
Please wait while we load your content...