Selective hydrogenation of CO2 over a Ce promoted Cu-based catalyst confined by SBA-15†
Abstract
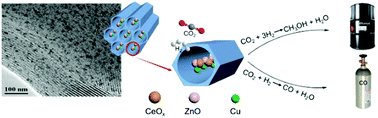
The chemical transformation of CO2 not only mitigates CO2 released into the atmosphere but also produces fuels and chemicals with important additional value. The activation of stable CO2 molecules is the key difficulty for the efficient conversion of CO2. Herein, we designed a ternary catalyst containing Cu, ZnO, and CeOx and dispersed them in SBA-15 using incipient wetness impregnation. Interestingly, the catalytic performances were improved greatly when the related active components were dispersed inside and outside the pore channels of SBA-15 using the incipient wetness impregnation method. Methanol and CO were controllably produced on the interface of Cu and metal oxides (ZnO and CeOx) due to the synergistic effects between the three active components. The metal oxides play a more important role in CO2 conversion as well as the product selectivity compared to Cu. CeO2 is more efficient than ZnO in promoting the dispersion of Cu nanoparticles and methanol generation. The suitable pore channels of SBA-15 promote the mass transfer and heat transfer while the high specific surface area is favorable to the dispersion of active components, which will facilitate the production of methanol and CO. After continuous hydrogenation evaluation for 100 h, the catalytic performance of the catalyst was still very stable. This work provides a viable strategy to prepare a supported catalyst with highly dispersed active sites, and the catalyst exhibits an efficient catalytic effect on producing methanol and CO.


 Please wait while we load your content...
Please wait while we load your content...