MoOx-modified bimetallic alloy nanoparticles for highly efficient hydrogen production from hydrous hydrazine†
Abstract
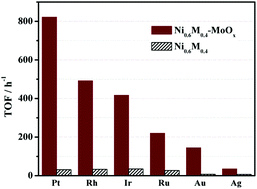
Hydrogen has been considered as an important alternative energy source for both environmental and economic reasons. The development of highly efficient and selective catalysts for hydrogen evolution from chemical hydrogen storage materials (e.g. hydrous hydrazine, N2H4·H2O) has become one of the most active research areas. In this study, MoOx-doped NiPt nanoparticles (NPs) have been successfully prepared via a simple one-step co-reduction strategy at room temperature. The characterization results show that the surfactant/support-free NiPt–MoOx NPs with a mean particle size of 5.0 nm are well-dispersed and in a low-crystalline/amorphous state. It is found that the prepared NiPt–MoOx samples show much higher catalytic activity toward the decomposition of N2H4 as compared to that of NiPt without the introduction of MoOx. In addition, among all the NiPt–MoOx NPs, the optimized Ni0.6Pt0.4–MoOx nanocatalyst shows 100% H2 selectivity and the best catalytic performance for the decomposition of N2H4 under alkaline conditions at 323 K. The TOF value of Ni0.6Pt0.4–MoOx NPs is found to be 822 h−1, which is among the highest values ever report for the same reaction. The excellent catalytic activities could be attributed to the modified electronic structure and the small particle size of NiPt NPs induced by the MoOx dopant. Other MoOx-doped Ni-based (NiM, M = Rh, Ir, Au, Ag and Ru) bimetallic catalysts also showed excellent catalytic activity. The present highly efficient and selective catalyst system may encourage the practical application of N2H4·H2O as a potential chemical hydrogen storage material.


 Please wait while we load your content...
Please wait while we load your content...