Influence of Zn and Co co-doping on oxygen evolution reaction electrocatalysis at MOF-derived N-doped carbon electrodes
Abstract
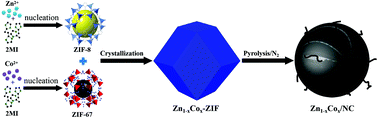
Electrochemical conversion of water into oxygen and hydrogen gas is a highly efficient method to meet the energy requirement on a global scale and solve the global warming problem arising from the mass consumption of fossil sources. Therefore, it is important to develop a catalyst with excellent oxygen reduction reaction (OER) activity. In this paper, we adopted the cocrystallization technology to synthesize Zn1−xCox-ZIF composites. Then, the Zn1−xCox-ZIF composites were used as precursors to prepare the Zn1−xCox/NC electrocatalyst (NC is N-doped carbon). The influence of the Zn element on the Zn1−xCox/NC electrocatalyst for the oxygen reduction reaction has been explored. All as-prepared samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), and X-ray photoelectron spectroscopy (XPS). The results show that Zn1−xCox/NC electrocatalysts exhibit excellent OER activity. And the doping of Zn has a great influence on the structure and OER efficiency of the Zn1−xCox/NC electrocatalyst. After the pyrolysis process, the organic ligands of Zn1−xCox-ZIF are transformed into porous carbon and Zn and Co nanoparticles are formed inside the carbon. Among these Zn1−xCox/NC electrocatalysts, Zn0.2Co0.8/NC shows the highest OER activity. The carbon nanowires appearing on the surface of Zn0.2Co0.8/NC nanoparticles are much more compared to that on others. The overpotential and Tafel slope of Zn0.2Co0.8/NC are 1.52 V and 98 eV dec−1, respectively. Zn0.2Co0.8/NC also exhibits outstanding long-term electrochemical durability. This finding can not only extend the application of MOFs, but can also present a new method for fabricating highly efficient OER electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...