Controlled aggregation of core(amorphous silica)@shell(TPA+-polysilicate) nanoparticles at room temperature by selective removal of TPA+ ions from the nanoparticle shell†
Abstract
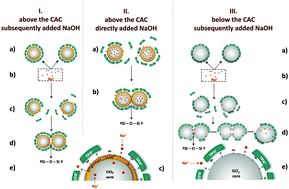
The influence of small inorganic cations (Na+ ions in this case) on core@shell silica nanoparticles, formed by the room temperature hydrolysis of TEOS in aqueous TPAOH (subsequent addition of NaOH – SA) and NaOH/TPAOH (direct addition of NaOH – DA) solutions, was investigated by DLS, AFM, pH measurements, 29Si-NMR and 23Na-NMR. The obtained results showed that smaller Na+ ions displace some of the larger TPA+ ions from the shells of the core@shell primary silica nanoparticles (PNPs), by the exchange process. This causes exposure of TPA-free terminal –OH and –O− groups to the solution and thus enables the binding of the collided nanoparticles by ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O–Si
Si–O–Si![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) bonds (aggregation). The PNPs in the aggregates formed above the critical aggregation concentration (CAC) of silicate species are separated by the “bulky” shell (TPA-polysilicate anions) that prevents close contact between the nanoparticle cores and thus their coalescence. When Na+ ions are present during the formation of the nanoparticles, some of the Na+ ions are incorporated into the nanoparticle core. Removal of the TPA+ ions from the shells of the nanoparticles formed below the CAC enables close contact of the nanoparticle cores and thus their coalescence inside the aggregates. These findings show that the selective removal of the TPA+ ions from the nanoparticle shell enables the control of the composition and size of the precursor species and thus represents a good basis for the engineering of nanosized (alumino)silicate microporous and mesoporous materials.
bonds (aggregation). The PNPs in the aggregates formed above the critical aggregation concentration (CAC) of silicate species are separated by the “bulky” shell (TPA-polysilicate anions) that prevents close contact between the nanoparticle cores and thus their coalescence. When Na+ ions are present during the formation of the nanoparticles, some of the Na+ ions are incorporated into the nanoparticle core. Removal of the TPA+ ions from the shells of the nanoparticles formed below the CAC enables close contact of the nanoparticle cores and thus their coalescence inside the aggregates. These findings show that the selective removal of the TPA+ ions from the nanoparticle shell enables the control of the composition and size of the precursor species and thus represents a good basis for the engineering of nanosized (alumino)silicate microporous and mesoporous materials.


 Please wait while we load your content...
Please wait while we load your content...