An iron(iii) complex selectively mediated cancer cell death: crystal structure, DNA targeting and in vitro antitumor activities†
Abstract
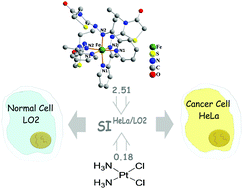
The design, synthesis, crystal structures and in vitro biological assessment of three iron(III) complexes, namely [Fe(L1)2]Cl (1), [Fe(L2)2]·FeCl4 (2) and [Fe(L3)(HL3)2]·(FeCl4)2·(CH3OH) (3) (HL1 = N’-(4-oxothiazolidin-2-ylidene)picolinohydrazonamide, HL2 = 2-(2-(1-(pyridin-2-yl)ethylidene)hydrazono)thiazolidin-4-one, HL3 = 3-ethyl-2-(2-(1-(pyridin-2-yl)ethylidene)hydrazono)thiazolidin-4-one, are reported in this article. Single crystal X-ray diffraction revealed that all three complexes were mononuclear molecules, approaching a coordination polyhedron of octahedral geometry, in which the 4-thiazolidinone derivative ligands exhibited different coordination modes and conformations. Their interactions with DNA were investigated by UV–visible and fluorescence spectrometries, and by agarose gel electrophoresis. Results indicated that they presented efficient DNA binding propensities and cleavage activities. In vitro, the iron(III) complexes caused cell cycle arrest at the G1 phase in response to DNA damage, effectively inhibited the cyclinD1 expression and decreased the migration of HeLa cells with a lower wound-healing rate than that of the control. Complex 3 was the most sensitive to HeLa cancer cells, followed by 2 and 1. Attractively, the tested complexes demonstrated significantly enhanced selectivity index values determined for HeLa vs. LO2 normal cells compared with cisplatin, especially for complex 3, which was up to 13.94-fold. Unlike cisplatin, more complex 3 would accumulate in HeLa cancer cells compared with LO2 normal cells.


 Please wait while we load your content...
Please wait while we load your content...