Multifunctional adenine-functionalized supramolecular micelles for highly selective and effective cancer chemotherapy†
Abstract
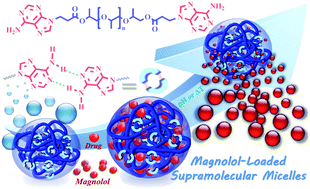
The facile construction of supramolecular polymers—incorporating self-complementary hydrogen bonds—with the chemical and physical properties required to achieve selective, safe, reliable drug delivery for chemotherapy remains highly challenging. In this study, we have successfully developed a new supramolecular polymer containing difunctional adenine-containing end groups that spontaneously self-assembles into nanospherical micelles in water and aqueous buffer solutions. The resulting supramolecular micelles exhibit good biocompatibility and tunable, reversible thermo-responsive phase transitions. Moreover, the drug-loading capacity and release behavior could be finely tuned using pH and temperature, making the micelles highly attractive potential candidates for controlled drug-delivery applications. Cytotoxicity assays and flow cytometric analyses clearly demonstrated that the drug-loaded micelles exhibited excellent drug entrapment stability and minimal cytotoxic effects against normal cells under normal physiological conditions. However, significantly increased intracellular uptake of the micelles—along with potent cytotoxic effects—was observed in cancer cells, indicating that the drug-loaded micelles could be selectively and rapidly endocytosed by cancer cells to induce tumor cell death without harming normal cells. Thus, this newly developed system has great potential as a selective tumor-targeted nanovector to improve the safety and efficacy of cancer chemotherapy.


 Please wait while we load your content...
Please wait while we load your content...