Studying the catalytic activity of DBU and TBD upon water-initiated ROP of ε-caprolactone under different thermodynamic conditions†
Abstract
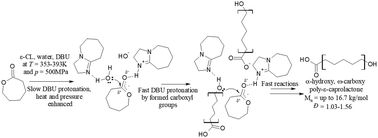
In this paper, catalyzed and non-catalyzed water-initiated Ring-Opening Polymerization (ROP) of ε-caprolactone (ε-CL) under various thermodynamic conditions (T = 293–393 K and p = 0.1–500 MPa) was carried out. As organocatalysts, two non-nucleophilic bases characterized by different modes of action, i.e. 1,8-diazabicyclo(5.4.0)undec-7-ene (DBU) and 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD), were used. High-pressure experiments revealed that polymerization proceeded much faster with respect to the reactions performed at ambient pressure (a limiting example of t = 168 h vs. t = 6 h) and the synthesized PCLs were characterized by molar masses, Mn, up to 16.7 kg mol−1 and low/moderate dispersity, Đ = 1.03–1.56. Moreover, as revealed by MALDI-TOF studies, polymers with telechelic characters were obtained. In each case the varying content of cyclic polyesters was detected as well. Their presence had a strong influence on the discrepancy between theoretically and experimentally determined Mn. Collected data surprisingly also revealed the enhanced catalytic activity of DBU over TBD at high pressure. One can add that for the ROP catalyzed by the latter compound, the termination process was still observed, while for a selected monomer to catalyst ratio a “pseudo-living” character of polymerization was warranted for the former catalyst. Further investigations with the use of FT-IR allowed us to hypothesize that TBD is fully protonated by water that is followed by cluster formation limiting its catalytic activity. On the other hand, the enhanced activity of DBU results from the presence of both forms (protonated and non-protonated) in reaction media acting as a Brønsted acid/base system. Our studies offer a new, more eco-friendly way to synthesize well-defined telechelic PCLs of moderate molecular weights in very short times and a better understanding of the role of intermolecular interactions in the catalytic activity of organocatalysts and generally the progress of ROP.


 Please wait while we load your content...
Please wait while we load your content...