Experimental model design: exploration and optimization of customized polymerization conditions for the preparation of targeted smart materials by the Diels Alder click reaction†
Abstract
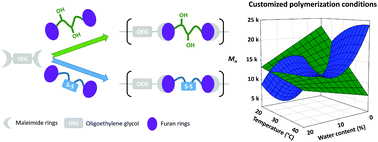
The experimental model design proposed herein has proved to be an indispensable tool to rapidly and easily elucidate the optimal polymerization conditions in the preparation of tailor-made responsive materials for biomedical applications. The use of reversible covalent bonds for the synthesis of new materials provides the polymer structures with novel functionalities and applications. The Diels–Alder (DA) reaction is an ideal synthetic method for the preparation of functional and responsive biomaterials under mild and undemanding conditions. The present work details the optimization of the polymerization conditions to synthesize two families of thermo-sensitive polymers, with either reduction-responsive properties or enhanced hydrophilicity. These polymers have been successfully prepared from two functional bisdiene monomers: a difurfuryl dithiodiethanol derivative (DiT-Fur, 1) and a difurfuryl-dithiothreitol derivative (DTT-Fur, 2), respectively, using the same bismaleimide (DMDOO, 3) derived from triethylene glycol in both systems. Twenty experiments were conducted and a Box–Behnken experimental design was used to evaluate the importance of temperature and the water content in terms of the polymer molecular weights (Mn, determined by 1H NMR). In addition, the progress of the polymerizations at selected times was accurately monitored via IR spectroscopy. Higher temperatures were discarded since previous thermo-lability studies demonstrated that the retro-DA reaction could occur at temperatures as low as 50 °C. Temperature proved to be the most influential variable in the final Mn, exerting opposite effects in both systems. We envision that this experimental approach, applied to other polymerization processes, can enlighten the influence of key parameters in the final product obtained.


 Please wait while we load your content...
Please wait while we load your content...