Charge reversal and swelling in saccharide binding polyzwitterionic phenylboronic acid-modified poly(4-vinylpyridine) nanoparticles†
Abstract
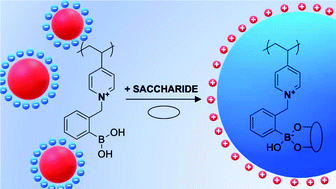
We present the synthesis and characterization of zwitterionic poly(4-vinylpyridine) nanoparticles modified with phenylboronic acid (QxPVP-PBA). We studied three different degrees of modification, which differed in both physicochemical and saccharide-sensing properties. The pKa values of QxPVP-PBA, determined by potentiometry and by 11B NMR spectroscopy, increased with the degree of PVP quaternization due to repulsion between charged groups. By dialysis of QxPVP-PBA polymers dissolved in HCl against MOPS buffer, we prepared stable nanoparticles at neutral pH, mostly stabilized by positive charges and by chains likely interconnected through cyclic PBA structures termed boroxines. QxPVP-PBA hydrophobicity increases with the decrease in the degree of PVP quaternization due to hydrophobic uncharged pyridine groups, as indicated by covalent binding to the PBA fluorescent probe alizarin. By potentiometric titrations and zeta potential measurements, we determined the pKa values of a fructose and glucose complex with the polymer, which were lower than the physiological pH. In addition, the alizarin displacement assay showed that QxPVP-PBA has a higher affinity to fructose than to glucose. Fructose binding leads to an increase in the size of the polymeric nanoparticles because their hydrophobicity increases after zwitterionic complex formation, as evaluated by LS and cryo-TEM. Notably, such fructose-bound particles showed a negative zeta potential. Therefore, QxPVP-PBA has unique charge reversal properties in the presence of saccharides.


 Please wait while we load your content...
Please wait while we load your content...