Simple Lewis pairs of zinc salts and organobases as bifunctional catalysts for controlled ring-opening polymerization of O-carboxyanhydrides†
Abstract
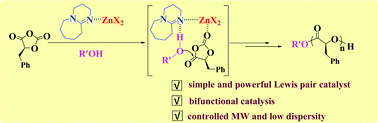
Simple Lewis pairs consisting of organobases (such as 1,8-diazabicycloundec-7-ene (DBU) and 7-methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene (MTBD)) and zinc salts ZnX2 (X = OAc, Cl, Br, and I) were explored to promote the ring-opening polymerization (ROP) of phenyl O-carboxyanhydride (PheOCA) for preparing well-defined poly(α-hydroxyalkanoic acid)s (PAHA)s. These Lewis pairs were found to exhibit great activity towards PheOCA in the presence of benzyl alcohol (BnOH), and the polymerizations produced linear PAHAs having predictable molecular weights, narrow molecular weight distributions (Mw/Mn < 1.1) and good isotacticity (Pm = 0.88). When hexanol, trimethylol propane (TMOP), pentaerythritol (PT) and hydroxyl-terminated poly(ethylene glycol) (such mPEGOH, HOPEGOH) were used, diverse chemical architectures including three-armed and four-armed PAHAs and even diblock and triblock amphiphilic polymers could be successfully fabricated. Careful analysis of the catalytic system verified the interaction between Zn(OAc)2, DBU and BnOH. Based on the kinetic experiments, it was evidenced that alcohols as initiators participated in the polymerization initiation, and the polymerization process was plausibly achieved via bifunctional catalysis of Lewis pairs, in which the monomers were activated through coordination to ZnX2, and the nucleophilicity of initiators promoted by organobases was beneficial for attacking the monomers for ring-opening. A set of catalytic systems consisting of metal salts and organobases is a powerful alternative to enhance the activity and selectivity of OCA polymerization for the well-defined synthesis of PAHAs.


 Please wait while we load your content...
Please wait while we load your content...