The synthesis, self-assembly and pH-responsive fluorescence enhancement of an alternating amphiphilic copolymer with azobenzene pendants†
Abstract
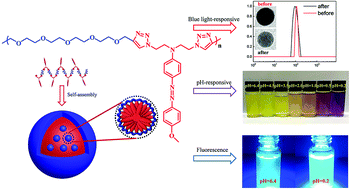
In this work, a novel alternating amphiphilic copolymer (AAC) P(EG4-a-NAzoOMe) with azobenzene pendants was synthesized through the azide–alkyne click reaction. This proposed AAC exhibits unusual photo- and pH-responsive behaviors by taking advantage of a special alternating amphiphilic topology to prevent the azobenzene pendants from ordered aggregation. P(EG4-a-NAzoOMe) could self-assemble into different sized homogeneous large compound micelles (LCMs) by tuning the initial concentration. The LCMs responded to UV stimuli almost as fast as the P(EG4-a-NAzoOMe) polymer, and the size of LCMs enlarged with the acidity of the solution together with the color change from yellow to violet. Moreover, the LCMs featured acidity-enhanced aggregation induced emission (AIE) similarly because of the special alternating amphiphilic topology. The first reported stimuli-responsive AAC could not only serve as a promising tunable delivery carrier or sensor, but also be inspirational for developing azobenzene AIE materials.


 Please wait while we load your content...
Please wait while we load your content...